Respiratory Panel
We have the best tests to help you prepare for the upcoming uncertainty.
Provista Diagnostics’ state-of-the-art, high-complexity clinical laboratory is accredited by the College of American Pathologists (CAP) and the Clinical Laboratory Improvement Amendments (CLIA). In response to the COVID-19 emergency, we have focused on delivering high-quality, fast turnaround COVID-19 testing in support of healthcare professionals, testing labs, policy-makers and the communities we serve.

Positive COVID-19 qPCR test will be used to
determine specific variant.
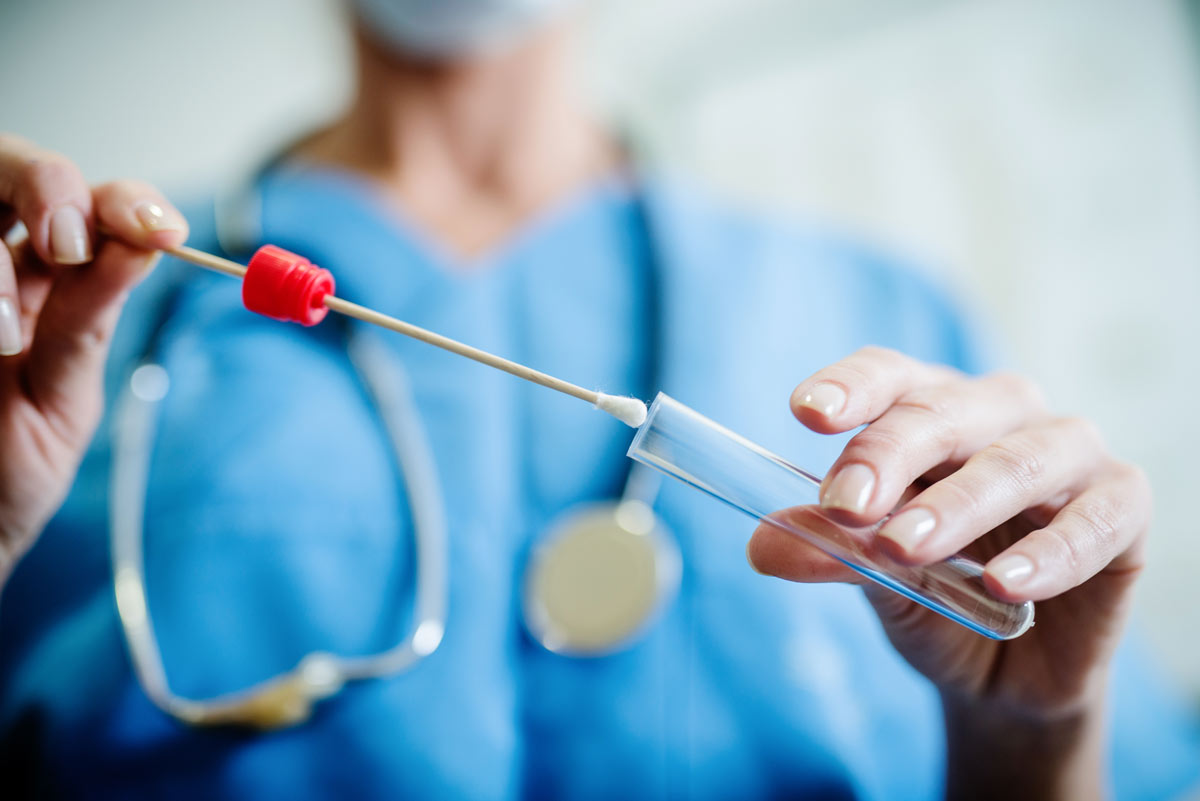
Collection Methods
We have validated, FDA-EUA approved Real time qPCR tests for any collection method:
-
Saliva: Collected by patient with or without supervision
-
Nasopharyngeal: Collected by healthcare provider
-
Oropharyngeal: Swabs the tonsil region of your mouth
-
Nasal: Extended swabbing at the entrance of your nose
-
Mid-Turbinate: Stops at nostril depth, deeper than a nasal swab
Available Tests:
-
COVID-19 qPCR: With and without FLU A/B and RSV; Identifies Delta and Delta Plus variants
-
Respiratory Viral: Multiplex Panel, Real-time qPCR assay, FluA/B, RSV, hMPV
-
Variant Identification: Positive COVID-19 test will be tested to determine specific variant; Identifies Delta and Delta Plus variants; Requires additional 24 hrs

